
We use mass number in naming isotopes, like Carbon-12 or Oxygen-17. Mass Number is the number of protons and neutrons in an isotope.From these measurements the average atomic mass can be calculated. Because isotopes for an element have different masses, they can be separated, and the percentage of each isotope measured. This laboratory instrument works based on the differences in the mass of the different isotopes of an element. To find the average atomic mass experimentally a mass spectrometer is used. The exact mass of an isotope is something that is determined experimentally. The answer, 12.011 amu, is the same value found for Carbon on the periodic table.Įxercise: Calculate the average atomic mass of Nitrogen (N) based on the information given. 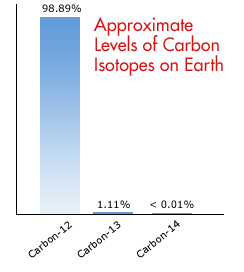
The table below shows the exact mass of each isotope (isotopic mass) and the percent abundance (sometimes called fractional abundance) for the primary isotopes of Carbon. To find the average atomic mass of the element Carbon, we multiply the mass of each isotope by its percent abundance expressed as a decimal. Like Carbon, many elements exist in nature as a mixture of isotopes. For example, if we take a weighted average for the isotopes of Carbon, we get an average atomic mass of 12.011 amu. The periodic table only tells us a weighted average of the atomic masses of the different isotopes for an element. The Periodic Table and Isotopes (Abundance)
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
